Kemiwatt, a French company founded in 2014, is developing its technology of Aqueous Organic RedOx Flow battery (AORFB).
RedOxFlow batteries (RFB) are an energy storage technology where chemical energy is provided by two chemical components dissolved in liquids to form electrolytes. As shown in Figure 1, during charge and discharge processes, electrolytes flow through a reactor called a cell stack. Redox reactions occur at the anode and cathode sides and electrolytes are respectively called “negolyte” and “posolyte”. The stack consists of several electrochemical cells stacked electrically in series using bipolar plates. Each cellcontains two electrodes that are separated by an ion exchange membrane.
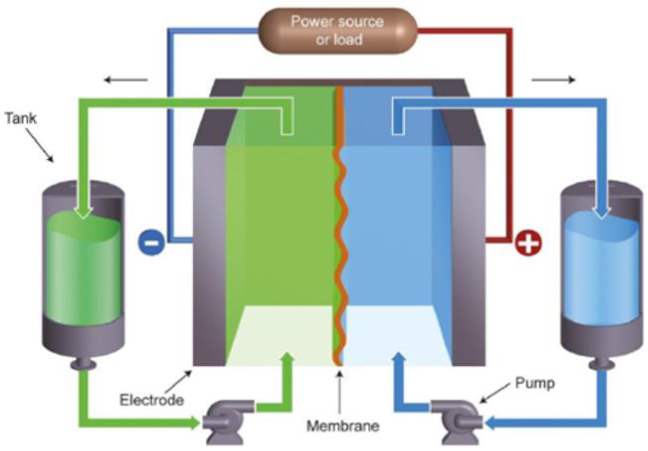
One of the main advantages of this technology is the decoupling ofpower and energy features, thus allowing flexibledesign. The energy capacity is determined by the volume of electrolyte stored in the external tanks while the power depends on the number of cells and their active surface area. Considering both a relative low power density and the amount of electrolyte flowing in the electrochemical stack beinga very few percent of the total electrolyte present, thus no-thermal runaway issue and a greater safety are offered by RedOx Flow batteries compared to other battery technologies. Also, the simplicity of electrode reactions (no need of noble metals as catalyst) contrasts with those of many conventional batteries (Lead-Acid, Sodium/Sulfur and Sodium/Metal-Halide, Li-Ion) involving phase transformations, electrolyte degradation or changes in electrode morphology. It ensures high lifetime and cyclability.
Considering the ability of releasing energy at constant power for 3 to more than 10 hoursand given its power density range, RedOx Flow Battery technologies are suitable for a wide range of energy services and stationary applications, such as:
- Integration of renewable energies from solar and wind farms
- Ancillary services and other network support services
- Microgrids and end-user energy optimization.
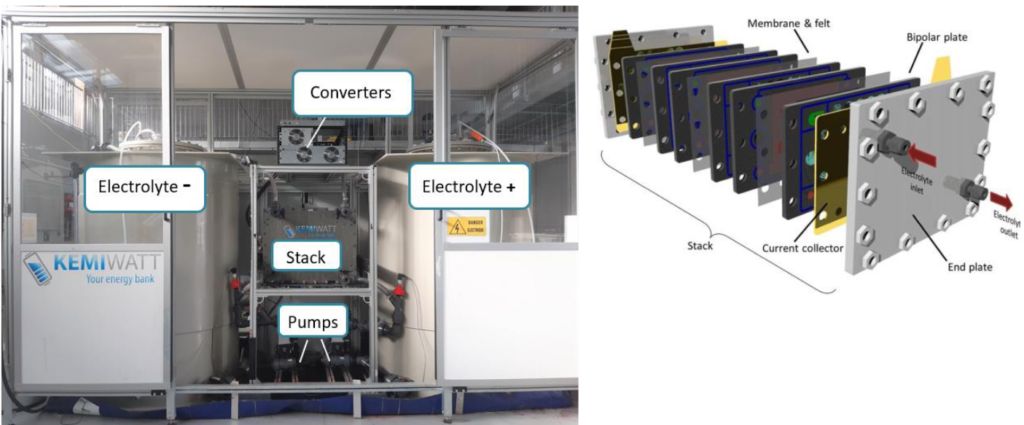
KemiwattElectrolytes are composed of potassium ferrocyanide, commonly used as food additive, in the posolyte and a proprietary anthraquinone derivative in the negolyte. Figure 2shows a picture of a fullAORFB system (at demonstrator scale) and the stack composition.
Regarding other RedOx Flow Battery technologies, the most mature and developed over the world is Vanadium RFB. However, this technology suffers from vanadium toxicity, its price fluctuation and a high corrosivity due to the necessity of using strong acidic electrolyte to dissolve this active speciesKemiwatt’ssystems are more environmentally friendly andcost-efficientdue to the use of organic molecules as active species which also areeasily recyclable. Kemiwatt’s AORFB also operatesin alkaline medium, less corrosive for stack components than vanadium RFB.
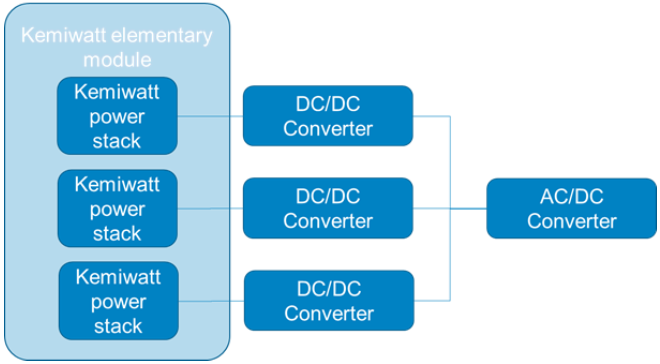
In order to ensure power from several kW to MW, Kemiwatt’ssystems may be composed of one or several stacks. In the last case, the electrical architecture ismodular and actuallybased onseveral DC/DC converters electrically in parallelcombined with only one low voltage AC/DC converterand thus limits electrical risk,as shownin Figure 3.This architecture induces two steps of conversion,each of them having their own efficiency, depending on power loading/unloading.
After a successful demonstration phase, the technology of Kemiwatt is currently under performance optimization along with industrialization, to start early-stage commercialization by this year.
Written by Guillaume Ozouf from Kemiwatt